  
The Shorthand electron configuration (or Noble gas configuration) as well as Full electron configuration is also mentioned in the table. 1999‑2023 - All Rights Reserved.Electron configuration chart of all Elements is mentioned in the table below. Īlso see: The Orbitron: A gallery of atomic orbitals and a few molecular orbitalsĬopyright © Israel Science and Technology Directory. The full story of the electron configurations of the transition elements. Thus, each proton and neutron has a mass of about 1 amu. This isotope of carbon has 6 protons and 6 neutrons. Atomic mass is measured in Atomic Mass Units (amu) which are scaled relative to carbon, 12C, that is taken as a standard element with an atomic mass of 12. Each element is uniquely defined by its atomic number.Ītomic mass: The mass of an atom is primarily determined by the number of protons and neutrons in its nucleus. Atomic number: The number of protons in an atom. The table below shows the full forms of the electron configurations of noble gases. For an explanation of these aspects, see the reference by Schwarz listed below. Thus, the electron configuration of Sc is 3d 1 4s 2. Starting with Scandium (Sc, atomic #21), the 3d orbital has a lower energy than the 4s. Energy levels and sublevels Principal energy level Therefore, orbital 4s is filled with electrons prior to orbital 3d. A step-by-step description of how to write the electron configuration for Arsenic (As and the As 3- ion).In order to write the As electron configuration we. The reason for this is that the energy level of orbital 4s is slightly lower than that of orbital 3d. Note that in the electron configuration of both K and Ca, the 4s orbital is filled before the 3d orbital. Since we had 6 electrons, we therefore have 6 protons, and the element with atomic number 6. The electron configuration of an atom of any element is the of electrons per sublevel of the energy levels of an atom in its ground state. The one additional electron configuration completes the picture for 19 electrons of Potassium. Electron Configurations of Atoms of Elements. The abbreviated form - 4s 1 - means the electron configuration of Argon (Ar), plus one electron in the 4s orbital. The full electron configuration of Potassium (K) is 1s 22s 22p 63s 23p 64s 1. Thus, the configuration shown for Potassium is 4s 1 (see Table below). Thus, substituting the config of He gives the full config for Neon: 1s 22s 22p 6įor example, for Potassium (K) (atomic #19), the preceding noble gas is Argon (Ar) (atomic #18). If you’re not familiar with the above terms, we recommend looking at Electron Shells to learn a bit more about them. For example, the abbreviated configuration for Neon is 2s 2 2p 6. Electron configuration, also known as electronic configuration, is the arrangement of electrons in shells, sub-shells, and orbitals within the atom. In the periodic table beyond Helium (He), each element's electron configuration is shown in an abbreviated form that starts with the symbol of the noble gas that precedes it. The superscript shows that there is one electron in the 1s orbital. In a neutral atom, the number of electrons equals the number of protons, which means. Its atomic number is 33, which is the number of protons in the nuclei of its atoms. The electron configuration of an element can be presented as a series of baseline and superscript symbols or it can be depicted schematically with a combination. Question: Is removing an electron from sodium or aluminium easier Answer: It does not take much energy. The simplest configuration is for Hydrogen: 1s 1. The complete electron for a neutral arsenic atom is: '1s'2'2s'2'2p'6'3s'2'3p'6'3d'(10)'4s'2'4p'3 Its shorthand electron configuration is: 'Ar''3d'(10)'4s'2'4p'3 As is the chemical symbol for the element arsenic. The electronic configuration of beryllium is 1s 2 2s 2. Now the goal of this video is to think about electron configurations for particular atoms. 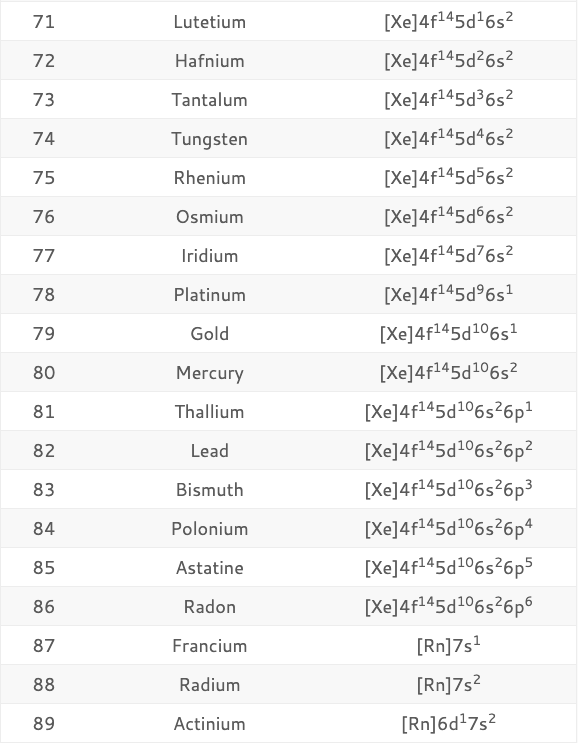
An atom's electron configuration describes the distribution of its electrons in the atomic orbitals ordered by the orbitals' energy levels. electronic configuration, also called electronic structure or electron configuration, the arrangement of electrons in orbitals around an atomic nucleus. The electronic configuration of N3- is 2,8 which is the same as the electronic configuration of an atom of the next Noble gas (Group 18) element in the Periodic. So if youre thinking about the subshell, the s subshell could fit two electrons, the p subshell can fit six electrons, the d subshell can fit 10 electrons, and the f subshell can fit 14 electrons, two per orbital.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
